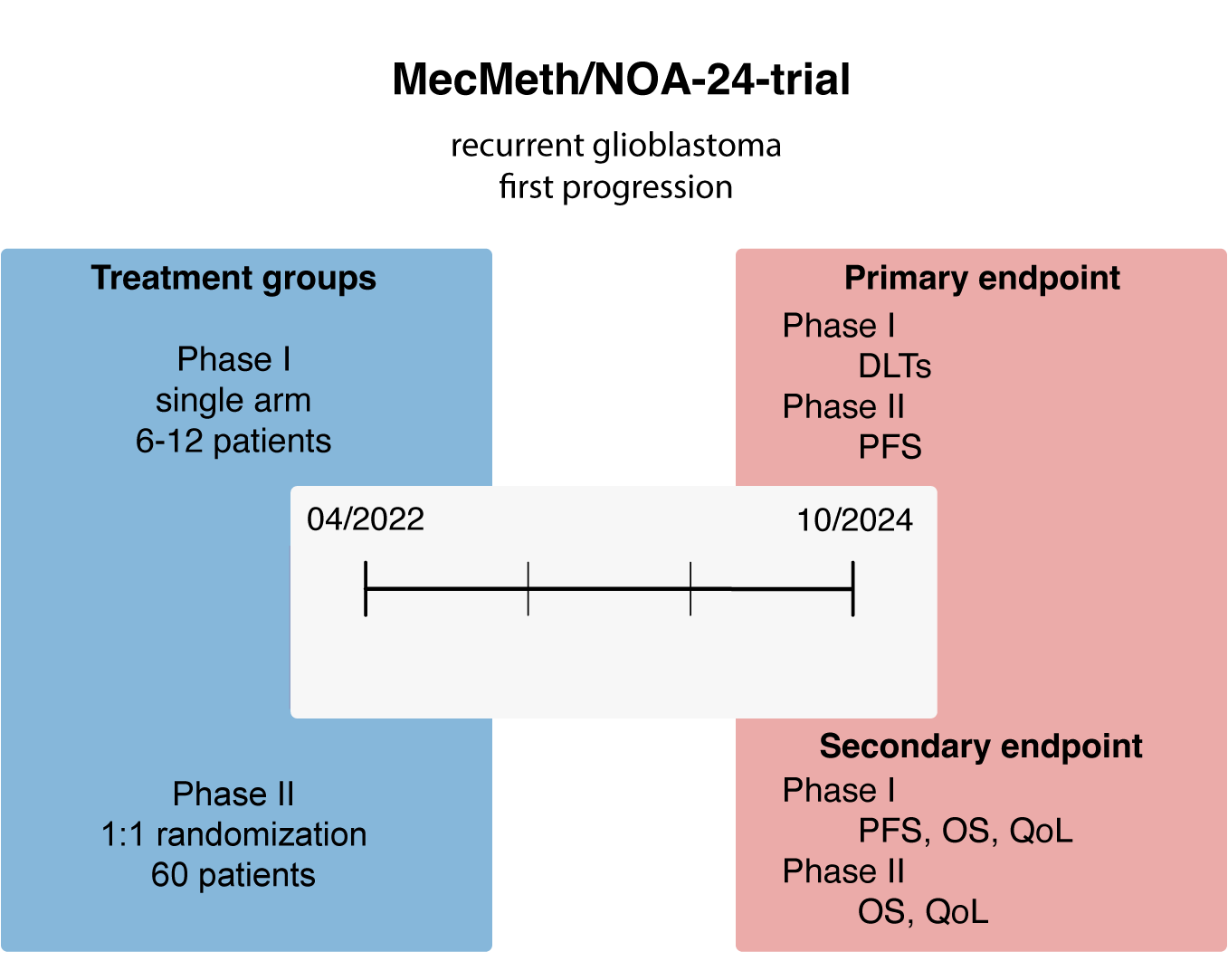
MecMeth/NOA-24 trial
Phase I/II trial of meclofenamate (MFA) in progressive MGMT-methylated glioblastoma under temozolomide second-line therapy
Objectives and endpoints
In the phase I component, the primary objective is to determine the toxicity of meclofenamate (MFA) therapy in addition to standard temozolomide (TMZ) in patients with the first relapse of MGMT promotor methylated glioblastoma. Efficacy, safety, tolerability and impact on quality of life are the secondary objectives. In phase II, the main objective is to determine the efficacy of MFA in addition to TMZ therapy. The primary endpoint in phase I is the incidence of dose-limiting toxicities (DLTs) during the first 8 weeks of MFA treatment. In phase II, the primary endpoint is progression-free survival (PFS) determined according to RANO criteria in cranial MRI.
Trial design
The study consists of a single-arm phase I part with 10 German trial sites and a randomized, parallel-group, and blinded phase II part with 15 German trial sites.
Inclusion criteria
- First relapse after first-line therapy with radiotherapy (RT) and alkylating chemotherapy, > 3 months after last chemotherapy application and >6 months after end of RT. Drug therapy and/or radiotherapy for first relapse treatment not yet started
- Tumor progression according to response-assessment in neurooncology (RANO) criteria
- Written informed consent
- Cognitive state to understand rationale and necessity of study therapy and procedures
- MGMT promotor-methylated, isocitrate-dehydrogenase (IDH) wildtype glioblastoma or gliosarcoma confirmed with histology of the primary resection
- Age > 18 years
- Karnofsky performance score (KPS) ≥ 60%;
- Life expectancy > 6 months
- Adequate bone marrow reserve (white blood cell count (WBC) >3 G/nl, platelets >100 G/nl)
- Adequate liver function (bilirubin <1.5 x upper limit of normal (ULN); ASAT /ALAT <3 x ULN, creatinine < 1.5 x ULN)
- Patient compliance and geographic proximity that allow adequate follow-up
- Male and female patients with reproductive potential must use an approved contraceptive method during and for 3 months after the trial (Pearl index <1%)
- Pre-menopausal female patients with childbearing potential: a negative serum pregnancy test (beta-HCG) must be obtained prior to treatment start
Additional criterion for phase I:
- Resection at first relapse not yet performed; according to the local treating neurosurgeon and the documented decision of local neurooncological tumor board, re-resection of the tumor is clinically indicated and can be safely deferred until day 7-10 after initiation of MFA/TMZ therapy.
Study protocol
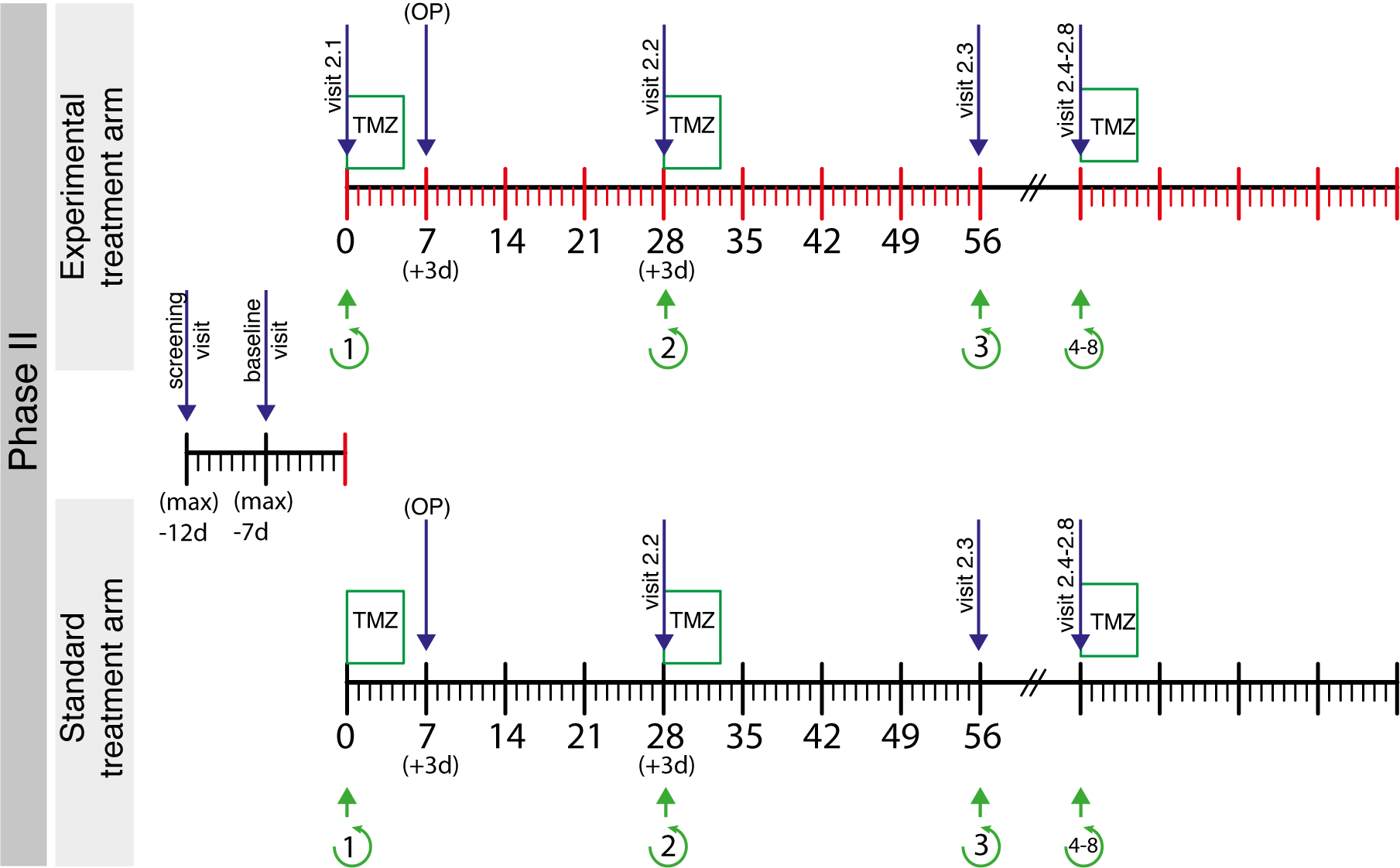
Modified from Zeyen, Potthoff et al., 2021, Trials
In phase I, there is one treatment arm with 6-12 patients. A dose-finding algorithm for MFA is performed. Patients are pretreated with MFA in addition to standard TMZ and tumor resection will be performed 7-10 days after therapy initiation. Postoperatively, TMZ is administered for eight 28-day cycles with daily MFA therapy.
Phase II will randomize 60 patients into an experimental treatment arm with MFA (dose determined in phase I) in addition to standard TMZ and the standard treatment arm with TMZ monotherapy. With 30 subjects per arm and a two-sided alpha level of 10%, this sample size provides a power of 80% at a hazard ratio of approximately 0.44, if a PFS-6 of 40% for the reference treatment is assumed. The trial is monitored by a Data Safety Monitoring Board.
Accompanying scientific program
As patients are pretreated with MFA 7-10 days prior to surgery in the phase I component, the resected tumor material provides the opportunity to perform MFA tissue level measurements and investigate the extent of blood-brain barrier permeability of MFA. Phase I and II will enable comprehensive morphological and transcriptional tumor network analysis and therefore provide human in-vivo insights into the cellular dynamics of a therapy that might target tumor microtube-based network connectivity.
Recruitment
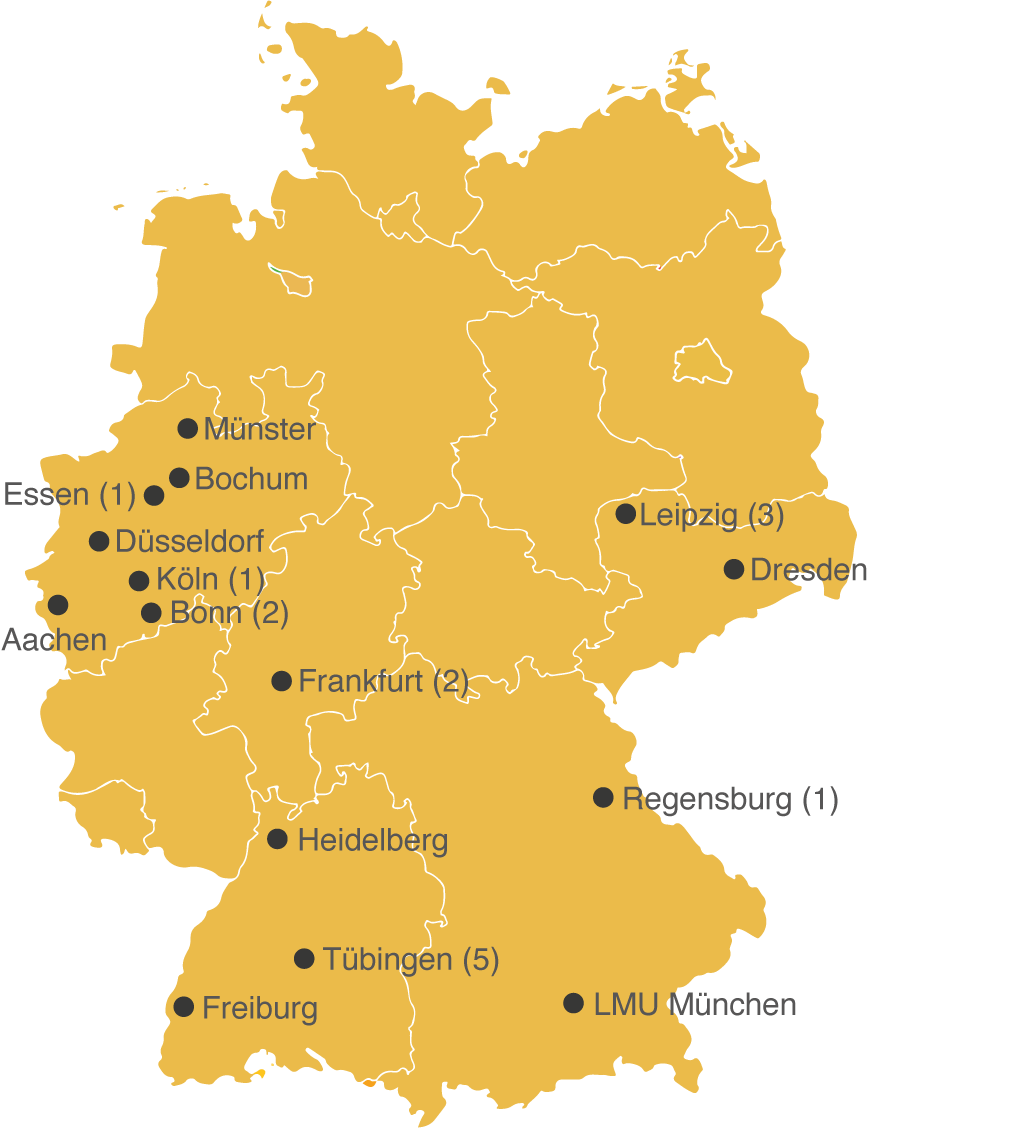
Initiation of the trial: April 2022
Number of patients included: 15
(Effective: 12 March 2025)
Funding

Trial registration
German Clinical Trials Register (DRKS): https://drks.de/search/en/trial/DRKS00025207/details;jsessionid=2442AF77D6F02F432187C206482B0634
EU Clinical Trials Register: https://www.clinicaltrialsregister.eu/ctr-search/trial/2021-000708-39/DE