Previous Clinical Trials
NOA-5 trial
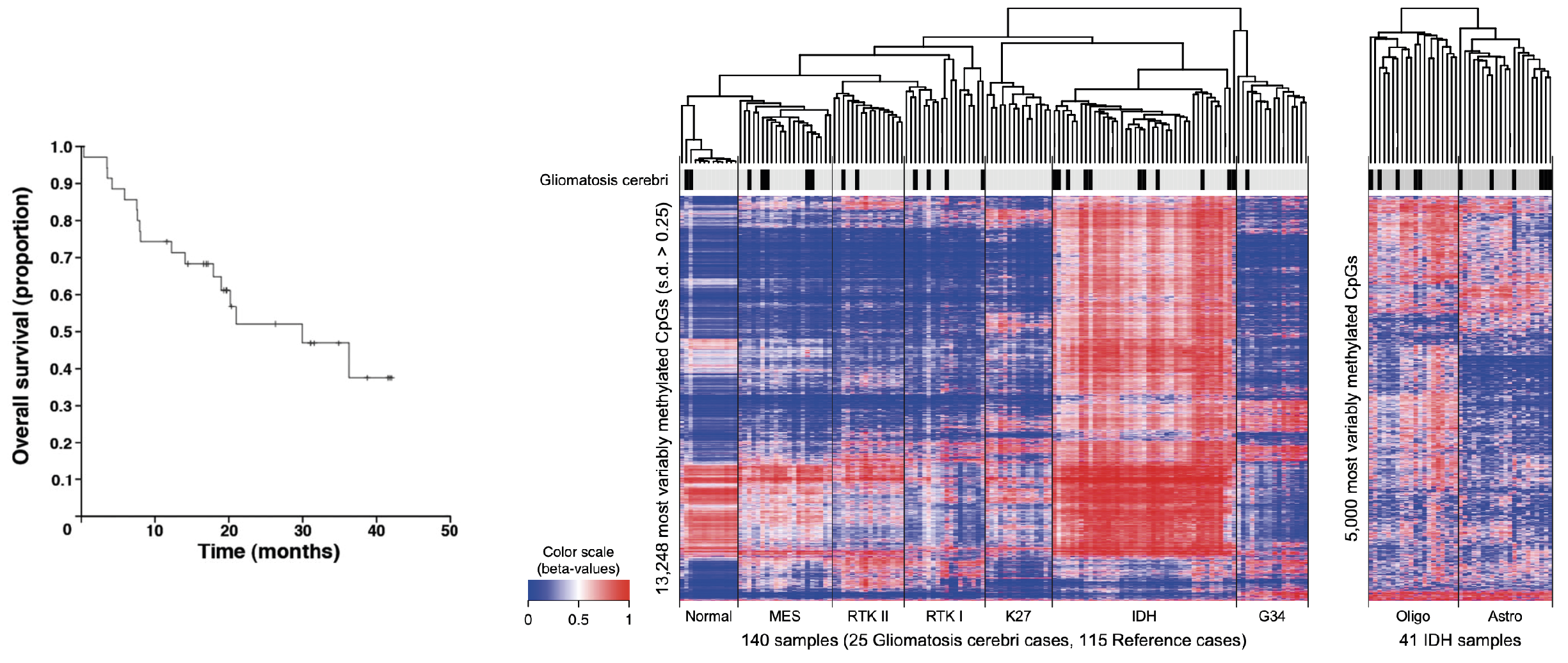
The NOA-05 trial for patients with gliomatosis cerebri established PC chemotherapy as a therapeutic option for gliomatosis cerebri (Glas et al., Ann Neurol 2011). Subsequent genetic analyses showed that, according to genetic alterations, gliomatosis cerebri cannot distinguished from gliomas grad II-IV raising raising doubt about gliomatosis cerebri being a separate brain tumor entity (Herrlinger et al., Acta Neuropathol 2016).
GLARIUS trial
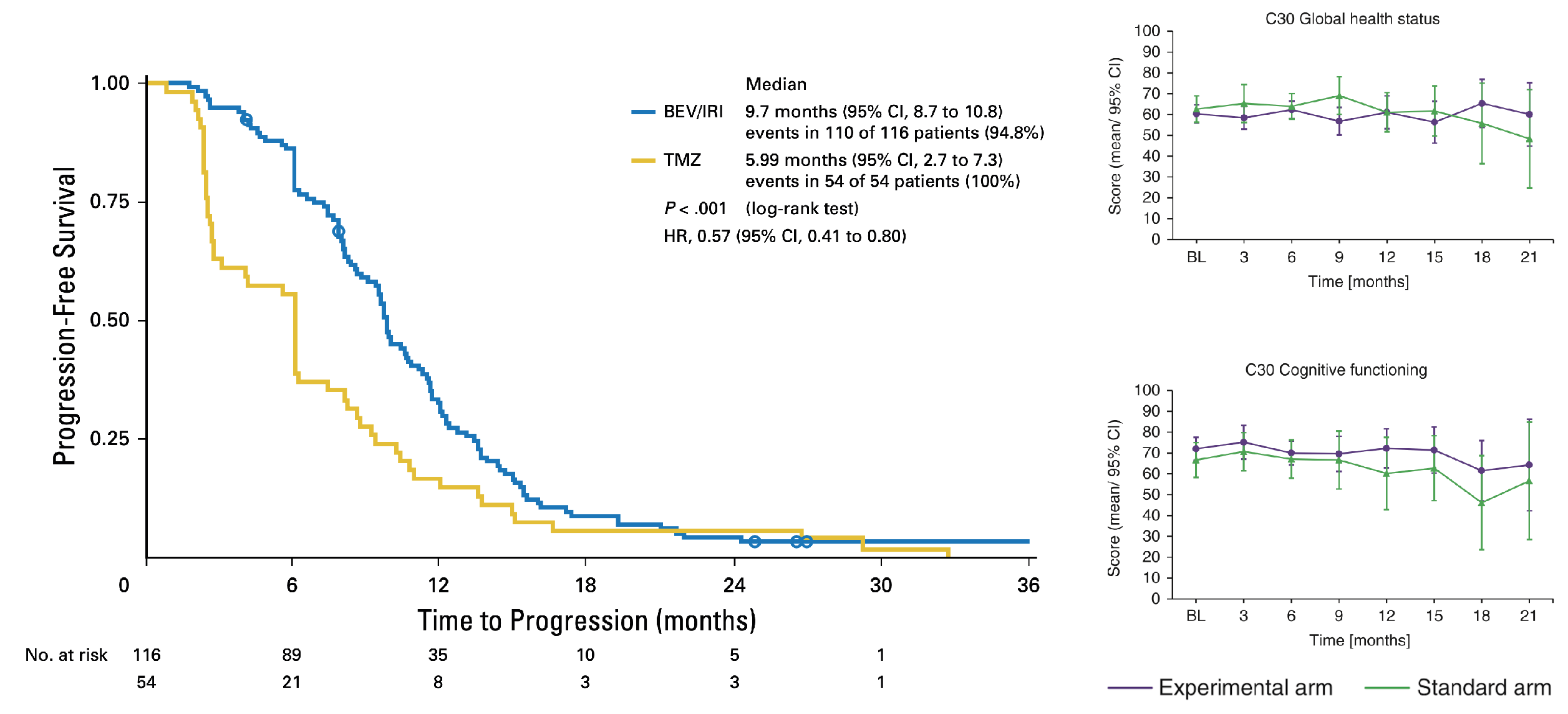
The GLARIUS trial showed PFS prolongation but no OS-prolongation for anti-VEGF-A-directed bevacizumab therapy in newly diagnosed MGMT-nonmethylated glioblastoma (Herrlinger et al., J Clin Oncol 2016). QoL analyses provided corroborative data supporting the notion that BEV may have beneficial effects upon QoL in relapsed GBM.
CeTeG/NOA-09 trial
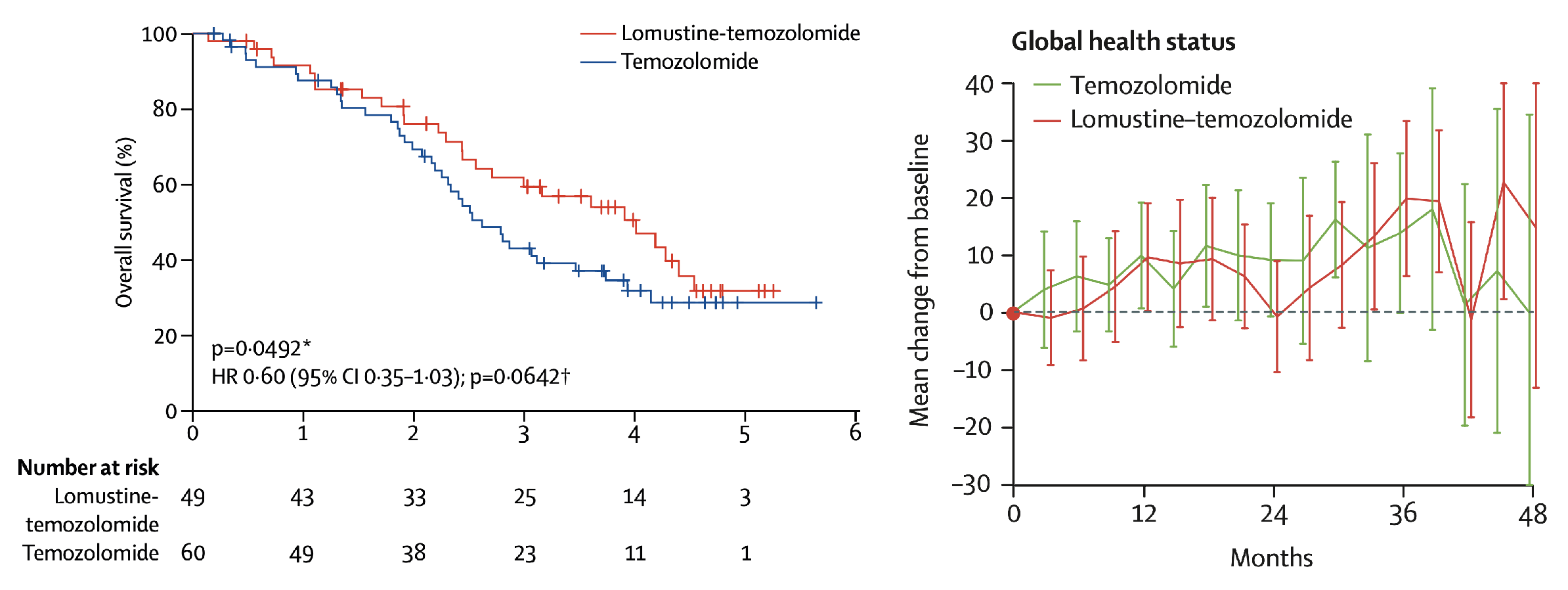
The CeTeG/NOA-09 trial showed that combined CCNU/TMZ chemotherapy ist superior regarding overall survival as comparted to TMZ standard therapy in newly diagnosed patients with MGMT-methylated glioblastoma (Herrlinger et al., Lancet 2019). In longitudinal analyses, the more intense CCNU/TMZ was not accompanied with a reduction in quality of life (QoL) or neurocognition (Weller et al., Lancet Oncol 2019).